Strategies to increase the intake and their effect on pregnant women
Introduction
LC-PUFA or long chain polyunsaturated fatty acids are organic acids containing more than 20 atoms of carbon and more than two double bindings, classified in n-3 PUFAs and n-6 PUFAs. From the physiological point of view, the most important LC-PUFAs are ARA: Arachidonic Acid (C 20:4 n-6) and DHA: Docosahexaenic Acid (C 22:6 n-3). Several researches have proven the importance of LC-PUFAs for pregnant women and newborn babies (Brenna, 2012).
Although the studies regarding supplementation with LC-PUFAs are sometimes controversial, most researches recommend a diet rich in fish, especially for pregnant women in the third trimester of pregnancy. During this stage of pregnancy LC-PUFAs are preferentially transferred to the developing foetus (Courville, et al., 2009). The purpose of this report is to analyse and determine which consumption form of LC-PUFA is best for pregnant women.
Physiological importance of LC-PUFAs for pregnant women
LC-PUFAs, notably DHA and AA, are crucial for the development of central nervous system, growth and body composition of the foetus. Furthermore, the accumulation of DHA in membranes of nervous cells is required for the development of retina and brain functions (Lagemaat, et al., 2011), while AA regulates foetal growth and is a precursor of eicosanoids which are important components of membrane lipids and have the role to mediate gene expression (Santos, et al., 2012; Hanebutt, et al., 2008).
Several studies have proven the acute importance of DHA during the third trimester of pregnancy (Guesnet and Alessandri, 2011), especially if the fact that pregnant women tend to have a diet deprived of DHA is considered (Courville, et al., 2009). The maternal intake of n-3 PUFAs, especially DHA (recommended average intake of 200-300 mg/day) will affect the DHA status of the newborn and its content in human brain phospholipids, and may also influence the health status in adult life, determining the vulnerability for chronic diseases and allergies and the reduction of cognitive capacity (Guesnet and Alessandri, 2011, Fernandes, et al., 2012). Moreover, DHA and other LC-PUFAs are crucial for immune and inflammatory responses and the adequate dietary supply will reduce the risk of preterm delivery (Hanebutt, et al., 2008).
Biosynthesis and pregnancy
Biosynthesis of LC-PUFAs should always be considered when proposing a diet choice for pregnant women, since the capacity of in utero production from precursors is limited and the levels of accretion are very high, particularly in the third trimester of pregnancy.
ARA and DHA are synthesized from dietary essential fatty acids (EFAs) such as linoleic acid (LA, 18:2 n-6) and α-linolenic acid (ALA, 18:3 n-3) by fatty acid desaturases and elongases in the maternal organism (Santos, et al., 2012). Image below by Kuipers et al.
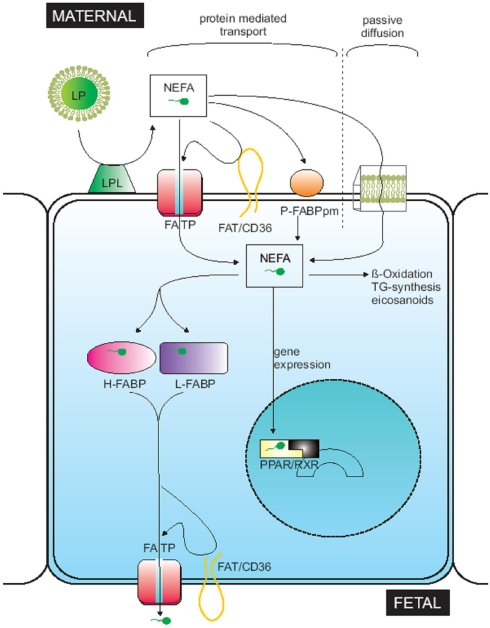
Given the limited capacity of the foetus to produce LC-PUFAs due to lack of desaturase activity, these will be transferred across the placenta from the maternal blood. The order of preference will be DHA > AA > ALA > LA (Hanebutt, et al., 2008). Santos et al., (2012) also report a priority of AA and DHA transfer from mother to foetus. The study suggests that the placenta has a higher selectivity for LC-PUFAs than for EFAs (Santos, et al., 2012). The accumulation of the DHA during cell maturation from the 6th month of pregnancy and during first months of birth and a simultaneous increase of absolute content of AA happens at very fast rates. The accretion of LA, AA and DHA will occur in skeletal muscle, skin, liver brain and adipose tissue in the order LA (342 mg/day)>AA (42 mg/day)>DHA (42 mg/day) at 5 weeks of gestation (Kuipers, et al., 2012). Table below by Kuipers et al. (2012).
Supplementation and changes in diet
Both n-3 and n-6 PUFA which come from maternal diet are important for the accretion of DHA and AA in membrane phospholipids of the developing foetus brain. Still, it has been proven that the Western diet is not optimal when considering the ratio of n-6/n-3, which exceeds the recommended value of 5 (Kouba and Mourot, 2011). A higher accretion of DHA was observed in East-African babies, the mothers of which have a diet lower in oils and higher in fish (Kuipers, et al., 2012). Moreover, a recent study demonstrated that diets deficient in DHA (even with an adequate intake of the precursor ALA) in the last 5 months of pregnancy can lead to lower visual acuity at 60 days of age (Guesnet and Alessandri, 2011). Several theories suggest that the increase of n-6 PUFAs in the diet has caused the raise of immune system dysfunctions (Helmersson-Karlqvist, et al., 2012), which resulted in augmentation of atopic diseases in developed countries (Krauss-Etschmann, et al., 2008).
Authors describe several ways to increase the intake of n-3 PUFAs. The most popular are:
- Changes in diet
- Diet rich in n-3 PUFA (fish or meat from animals with diets rich in n-3 PUFA) (Fernandes, et al., 2012)
- Diet rich in n-3 PUFA precursors
- Supplementation with fish oil
Thus, the question arises whether a change in the diet or supplementation with n-3 LC-PUFA from fish oil or other sources are necessary for pregnant women. Several studies were undertaken in order to determine the consequences of a higher intake of n-3 PUFAs by pregnant women. Some of them were focused on changes in diet, i.e. a higher intake of oily fish, while others regarded the effects of supplementation with fish oil. The purpose of both approaches was to study the effect on the content of n-3 PUFA in the foetus and the influence on certain physiological functions such as immunity.
The study of Helmersson-Karlqvist, et al., (2012) revealed that the consumption of farmed salmon by pregnant women twice per week (EPA and DHA, 403 mg/ day) affected the levels of fatty acids in pregnant women, but not the urinary 15-keto-dihydro-PGF2a which is a metabolite of PGF2a: a primary prostaglandin with a double role in reproductive endocrinology and inflammation. Thus, the consumption of oily fish up to two times per week will not have adverse effects on pregnancy, labour and parturition. Whereas, supplementation with fish oil proved to have influence on eicosanoid production, presumably due to intake of higher doses of DHA and EPA. (Helmersson-Karlqvist, et al., 2012).
Furthermore, experiments on the diets of rats demonstrated that consumption of products rich in precursors of n-3 PUFA can result in the same blood levels of n-3 PUFA as in the case of intake of products containing ready-made n-3 PUFA. Moreover, the influence of the diet on the immune markers was studied, revealing a direct effect of fish and linseed oil consumption on the differentiation of cells within foetal thymus (Childs, et al., 2010). However, the facts that the accretion rates are very high in the last trimester of pregnancy and that the capacity of the foetus for synthesis from precursors is limited should be considered when manipulating the diet.
Other studies regarding fish consumption by pregnant women demonstrate that mothers with low intake of fish increase the risk of sub-advancement of fine motor skills and social developmental scores (Hanebutt,et al., 2008). Another question regarding altered ingestions of n-3 and n-6 PUFAs is their effect on modulation of allergic diseases, since allergies are stimulated in utero and early infancy. A European multi-centre research including pregnant women from Spain, Germany and Hungary, the diet of which was supplemented with fish oil, showed a decrease in mRNA levels of TH2 related molecules in the foetus and in maternal inflammatory cytokines, which may have as consequence the declining incidence of allergies in children. Also, a study from Norway showed that consumption of n-3 LC-PUFAs from cod liver during pregnancy will enhance mental development of the offspring (Hanebutt, et al., 2008), while the study of Decsi (2009) demonstrated that supplementation with n-3 PUFA extends the gestation term. Nevertheless, other studies showed that fish oil supplementation of pregnant women may also have negative results such as higher incidence of eczema (Krauss-Etschmann, et al., 2008).
Conclusions
Both n-3 and n-6 LC-PUFAs are important for the development of nervous and immune systems and growth of the fetus, but DHA becomes crucial from the 6th month of pregnancy
Limited in utero synthesis and high accretion rates result in vital importance of LC-PUFA in the diet of pregnant women
From the several methods to increase the intake of LC-PUFAs, notably n-3 PUFAs, the most suitable would be the high consumption of oily fish or fish liver. High intake of fish proved to be a safer choice compared to fish oil supplementation.
Bibliography
Brenna, T (2012) Tissue-specific LCPUFA accretion in fetal humans, Prostaglandins,Leukotrienes and Essential Fatty Acids , 1.
Childs, C E, Hoile, S P, Burdge, G C and Calder, P C (2012) Changes in rat n-3 and n-6 fatty acid composition during pregnancy are associated with progesterone concentrations and hepatic FADS2 expression, Prostaglandins, Leukotriene sand Essential Fatty Acids , 141-147.
Childs, C E, Romijn, T, Enke, U, Hoile, S and Calder, P C (2010) Maternal diet during pregnancy has tissue-specific effects upon fetal fatty acid composition and alters fetal immune parameters, Prostaglandins, Leukotrienes and Essential Fatty Acids , 179-184.
Courville, A, Keplinger, M R, Judge, M P and Lammi-Keefe, C J (2009) Plasma or red blood cell phospholipids can be used to assess docosahexaenoic acid status in women during pregnancy, Nutrition Research , 151-155.
Decsi, T (2009) Effects of Supplementing LCPUFA to the Diet of Pregnant Women: Data from RCT, Advances in Experimental Medicine and Biology , 65-69.
Fernandes, F S, Sardinha, F L, Badia-Villanueva, M, Carulla, P, Herrera, E and Tavares do Carmo, M G (2012) Dietary lipids during early pregnancy differently influence adipose tissue metabolismand fatty acid composition in pregnant rats with repercussionson pup’s development, Prostaglandins, Leukotrienes and Essential Fatty Acids , 167-174.
Guesnet, P and Alessandri, J M (2011) Docosahexaenoic acid (DHA) and the developing central nervous system (CNS). Implications for dietary recommendations, Biochimie , 7-12.
Hanebutt, F L, Demmelmair, H, Schiessl, B, Larque, E and Koletzko, B (2008) Long-chain polyunsaturated fatty acid (LC-PUFA) transfer across the placenta, Clinical Nutrition , 685-693.
Helmersson-Karlqvist, J, Miles, E A, Vlachava, M, Kremmyda, L S, Noakes, P S and Diaper, N D (2012) Enhanced prostaglandin F2a formation in human pregnancy and the effect of increased oily fish intake: Results from the Salmon in Pregnancy Study, Prostaglandins, Leukotrienes and Essential Fatty Acids , 35-38.
Kouba, M and Mourot, J (2011) A review of nutritional effects on fat composition of animal products with special emphasis on n-3 polyunsaturated fatty acids, Biochimie , 13-17.
Krauss-Etschmann, S, Hartl, D, Rzehak, P, Heinrich, J, Shadid, R and Ramı´rez-Tortosa, M C (2008) Decreased cord blood IL-4, IL-13, and CCR4 and increased TGF-b levels after fish oil supplementation of pregnant women, Journal of Allergy and Clinical Immunology , 464-470.
Kuipers, R S, Luxwolda, M F, Offringa, P J, Boersma, E R, Dijck-Brouwer, D A and Muskiet, F A (2012) Fetal intrauterine whole body linoleic, arachidonic and docosahexaenoic acid contents and accretion rates, Prostaglandins ,Leukotrienes and Essential Fatty Acids , 13-20.
Lagemaat, M, Rotteveel, J, Muskiet, F, Schaafsma, A and Lafeber, H N (2011) Post term dietary-induced changes in DHA and AA status relate to gains in weight, length and head circumference in preterm infants, Prostaglandins, Leukotrienes and Essential Fatty Acids , 311–316.
Santos, F S, Chaves, C R, Costa, R S, Oliveira, M S, Santana, M G and Conceisao, F D (2012) Status of cis and trans Fatty Acids in Brazilian Adolescent Mothers and Their Newborns, Journal of Pediatric and Adolescent Gynecology , 270-276.


